SYSTEMS BIOLOGY OF
MAMMALIAN TISSUES
SYSTEMS BIOLOGY OF
MAMMALIAN TISSUES

Address
Room 159, Nella & Leon Benoziyo Building for Biological Science, Department of Molecular Cell Biology, Weizmann Institute of Science Rehovot 76100, Israel
Telephone
+972-8-934 3104
Fax
+972-8-934 4125

The mammalian liver performs critical functions for maintaining metabolic homeostasis. These functions are carried out by hepatocytes operating in repeating anatomical units termed liver lobules. The liver lobule is polarized by centripetal blood flow, creating gradients of nutrients, hormones and oxygen, and as a result hepatocytes at different lobule coordinates sub- specialize in distinct functions. We use single molecule imaging methods and single-cell genomics to study how this spatial division of labor within the liver lobule can serve to bring about optimal tissue function. We also study its impact on liver disease and liver regeneration.
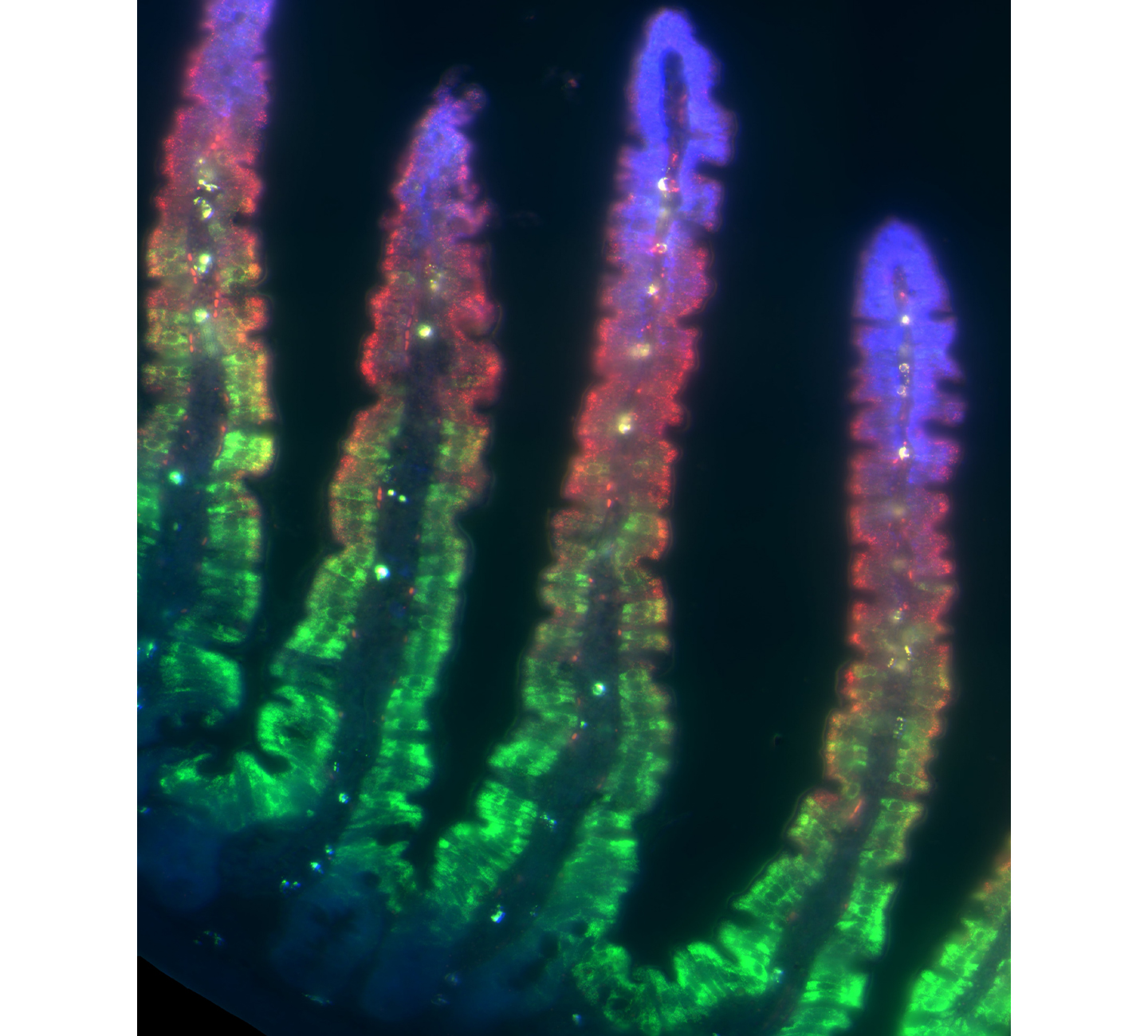
Our intestines are lined with a single layer of epithelial cells that forms a highly folded structure. Cells are constantly emerging from deep stem-cell harboring crypts and migrate along the walls of protrusions called villi until they reach their tips, where they are shed off into the lumen. Using spatially resolved single cell RNA sequencing, we found that epithelial cells constantly change their function as they migrate along the villi walls, specializing in distinct tasks at different villi heights. We study the design principles of these zonated expression programs, the mechanisms that shape them and their changes in diverse metabolic disease.
The human digestive tract sheds massive amounts of cells every day. We found that these cells remain viable and can be transcriptionally profiled to yield valuable information on human pathophysiology. We characterize shed cells in fecal matter to characterize major human digestive tract pathologies, including inflammatory bowel diseases, colorectal cancer and celiac disease.